























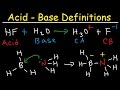














































Answer to Identify the conjugate acid-base pair in the equation: H3O+(aq)+CO32−(aq)⇌H2O(l)+HCO3−(aq) Identify the conjugate conjugate acid base pair differs by H+ . so HCO3- and CO32- is one such pair in which HCO3- is the acid and CO32- is the conjugate base. and another pair will be H2O and H3O+ ..in which H2O is the... Explanation. What is the conjugate base of HCO3-? Answer. The conjugate base of bicarbonate, HCO 3- is carbonate, CO3 2-. HCO3- is a conjugate acid, H 2 CO 3. Log in or register to post comments. A conjugate base contains one less H atom and one more - charge than the acid that formed it. Let us take the example of bicarbonate ions reacting with water to create carbonic acid and hydronium ions. HCO₃⁻ + H₂O → H₂CO₃ + OH⁻ base + acid → Conj A + Conj B We see that HCO₃⁻ becomes H₂CO₃. When the reaction is going to the left, HCO3- on the right side of the equation loses H+ to SO4-2, so HCO3- is acting as an acid. On the left side of the equation will be the conjugate base of HCO3- which must be CO3-2 or carbonate ion. Conjugate Acid: Bronsted-Lowry reactions describe the interactions of acids and bases in terms of the transfer of protons. An acid that donates a proton, usually in the form of H+, is called a For a, the acid is H2CO3, and the base is water. During the acid-base reaction, you remove a proton (or hydrogen atom) from H2CO3, and attach it to H2O. Therefore H2CO3 becomes HCO3-, and H2O... So, in order to determine the conjugate acid for a given base, all you have to know is that. In your case, the base is hydrogen carbonate, or H CO− 3. If you write the equation you'll get. Carbonic acid, or H 2CO3, will be the conjugate acid of hydrogen carbonate. View this answer. The conjugate base of HCO 3 - is CO 3 -2, which is the carbonate ion. To determine the conjugate base of a substance, you remove one hydrogen ion. It's... See full answer below. 1) What is the conjugate base of HCO3−? Express your answer as a chemical formula. 2) What is the conjugate acid of HPO32− ? Express your answer as a chemical formula. 3) Among three bases, X−, Y−, and Z−, the strongest one is Y−, and the weakest one is Z−. Rank their conjugate acids, HX, HY, and HZ, in order of decreasing strength.
[index] [6116] [7156] [1085] [8319] [6315] [7101] [2655] [5472] [5889] [6290]
Copyright © 2024 hot.realmoneygamestop.xyz